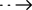SARS-CoV-2 & Influenza A/B & RSV Antigen Combo Rapid Test Kit (LFIA)
Medomics Flu & RSV Antigen Combo Rapid Test Kit (LFIA) : Respiratory tract infections are one of the most important causes of human illness and death. There are many pathogens that cause respiratory infections, including bacteria, viruses, and mycoplasma, with viruses in particular being the most common. Although the clinical signs and symptoms of respiratory tract infections are similar, the treatment of infections caused by different types of pathogens varies greatly, as do the efficacy and course of the disease; therefore, rapid and accurate detection and identification of pathogenic microrganisms can help in clinical diagnosis and treatment.
Intended use
Virus Flu & RSV Antigen Combo Rapid Test Kit (LFIA) is an immunochromatography based one step in vitro test. It is designed for the rapid qualitative determination of virus, Inflfluenza A , Inflfluenza B and RSV virus antigen in swab samples from individuals suspected of virus, Inflfluenza A , Inflfluenza B and RSV within the fifirst seven days of symptom onset. Virus & Inflfluenza A/B Antigen Combo Rapid Test Kit (LFIA) shall not be used as sole basis to diagnose or exclude virus, Inflfluenza A , Inflfluenza B and RSV infection.

*Test cassette: contains the virus & Flu & RSV Antigen test strip and a plastic cassette casing.Virus& Flu & RSV Antigen test strip contains anti-virus Nucleocapsid Protein antibody labeled with colloidal gold, anti-Inflfluenza A Nucleocapsid Protein antibody labeled with colloidal gold, anti-Inflfluenza B Nucleocapsid Protein antibody labeled with colloidal gold anti-RSV antibody labeled with colloidal gold. Another anti-virus Nucleocapsid Protein antibody, anti-Inflfluenza A Nucleocapsid Protein antibody, anti-Inflfluenza B Nucleocapsid Protein antibody and anti-RSV antibody are fixed on the N line, A line , B line and R line respectively. The N line/A line/B line / R line and control line (C line) are in the detection window on the nitrocellulose membrane.
Storage Instructions:
The test kit should be stored away from direct sunlight at 2ºC to 30ºC with a shelf-life of 24 months. Do not freeze.
Sample Requirements:
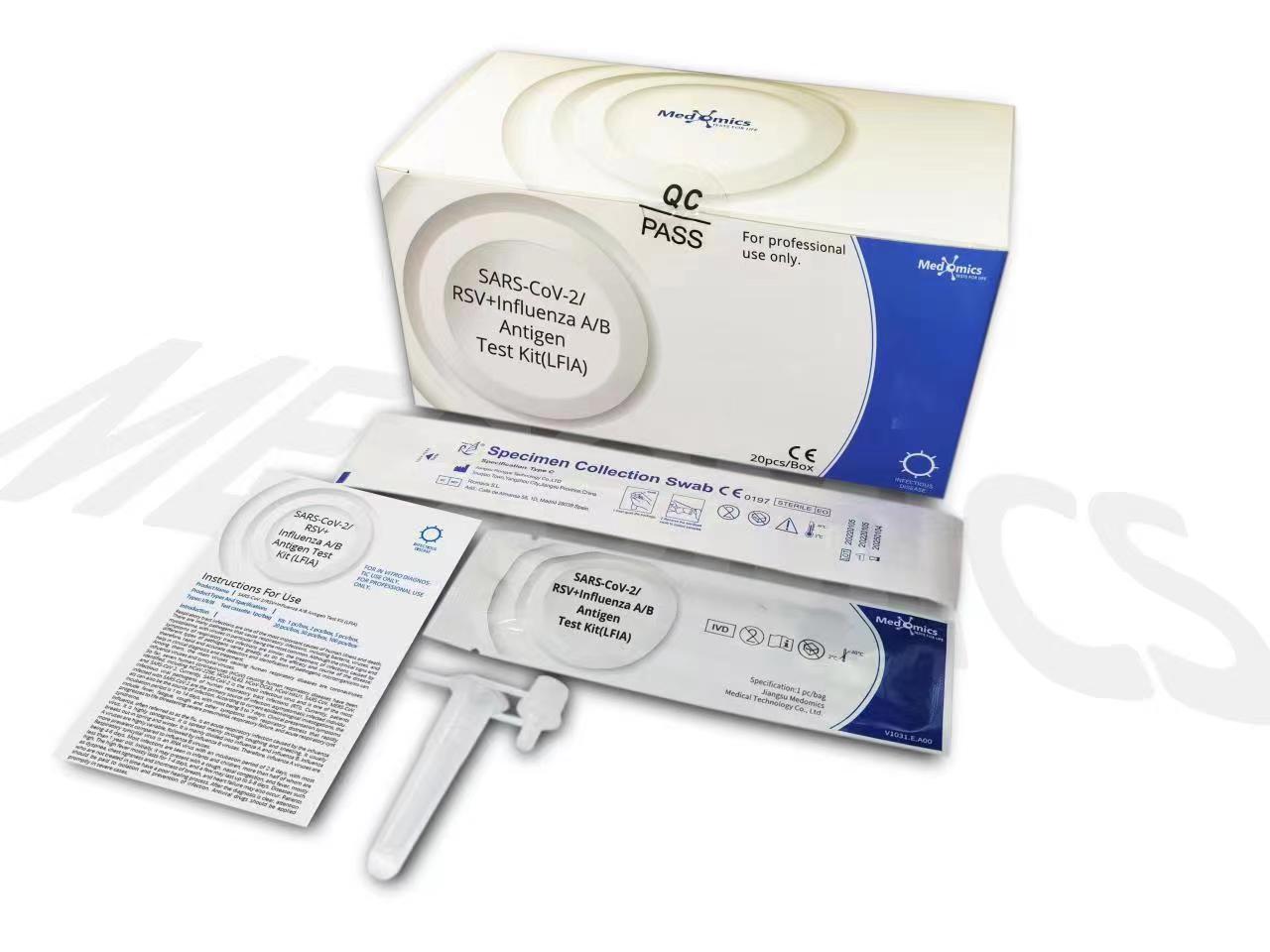
One test cassette can only be used to test one sample type. Sample types include anterior nasal secretion, nasopharyngeal secretion and throat secretion.
Sample should be treated with lysis buffer provided in this kit as soon as possible after collection. If the sample cannot be processed immediately, it should be stored immediately in a dry, sterilized and strictly sealed plastic tube. It can be stored at 2ºC-8ºC for 8 hours. Could be stored at -70ºC for long term storage.
Test Procedure:
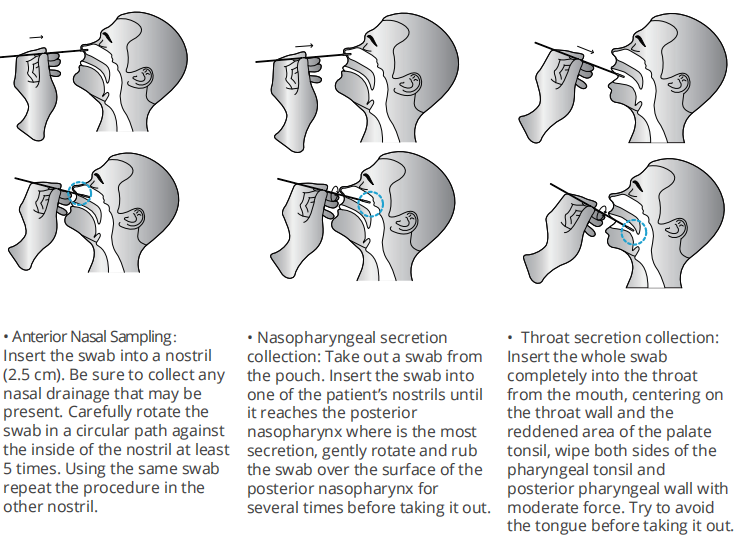
Do not open pouch until ready to use. Prep necessary materials: Timer | Tube rack for sampling tubes and specimens | Any necessary personal protective equipment. 1 | Sampling: Vertically add 15 drops (approximately 350 μL) lysis buffer into the sampling tube from vial or open the seal of the sampling tube containing lysis buffer or twist and squeeze out all the lysis bffer into the sampling tube from capsule. Insert the swab (after collection) into the buffer. Rotate the swab against the inner tube wall 10 times and squeeze the swab from the outer tube wall 5 times to completely dissolve the sample in the buffer, then move the swab up until it is resting on the sample solution, squeeze the swab from the outer tube wall in order to leave the sample in the tube as much as possible.
Remove and discard the swab, cover the tube with the dropper. 2 | Test procedures: Open the aluminum foil pouch, take out the test cassette and lay it on a clean flflat surface, then mark the cassette with the patient ID or sample number and add 4 drops (approximately 100 μL) processed sample extract into the sample well. The result should be observed within 15-20 minutes. Results observed after 20 minutes are invalid.
Certificate

- Email Us overseas@medomics-dx.com
- Call Us +86-025-58601060
- COVID-19 Solution
- Top
Contact Us
Phone: (+86) 025 - 58601060
E-mail: overseas@medomics-dx.com
Address:Building 01, Phase 6, No.71, Xinghui Road, Jiangbei New Area, Nanjing
- Email Us
- COVID-19 Solution
- Call Us (+86) 025 - 58601060
- top
Jiangsu Medomics Medical Technology Co,Ltd Powered By www.300.cn