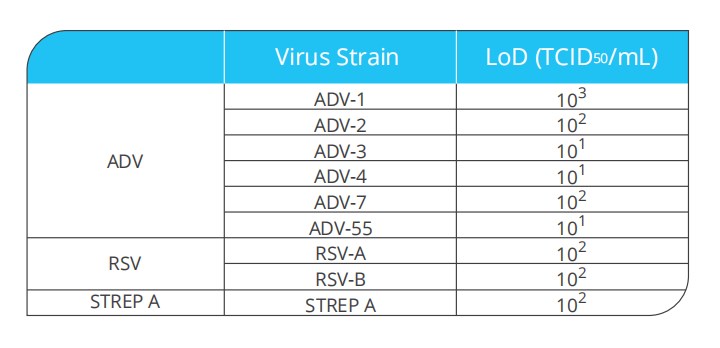
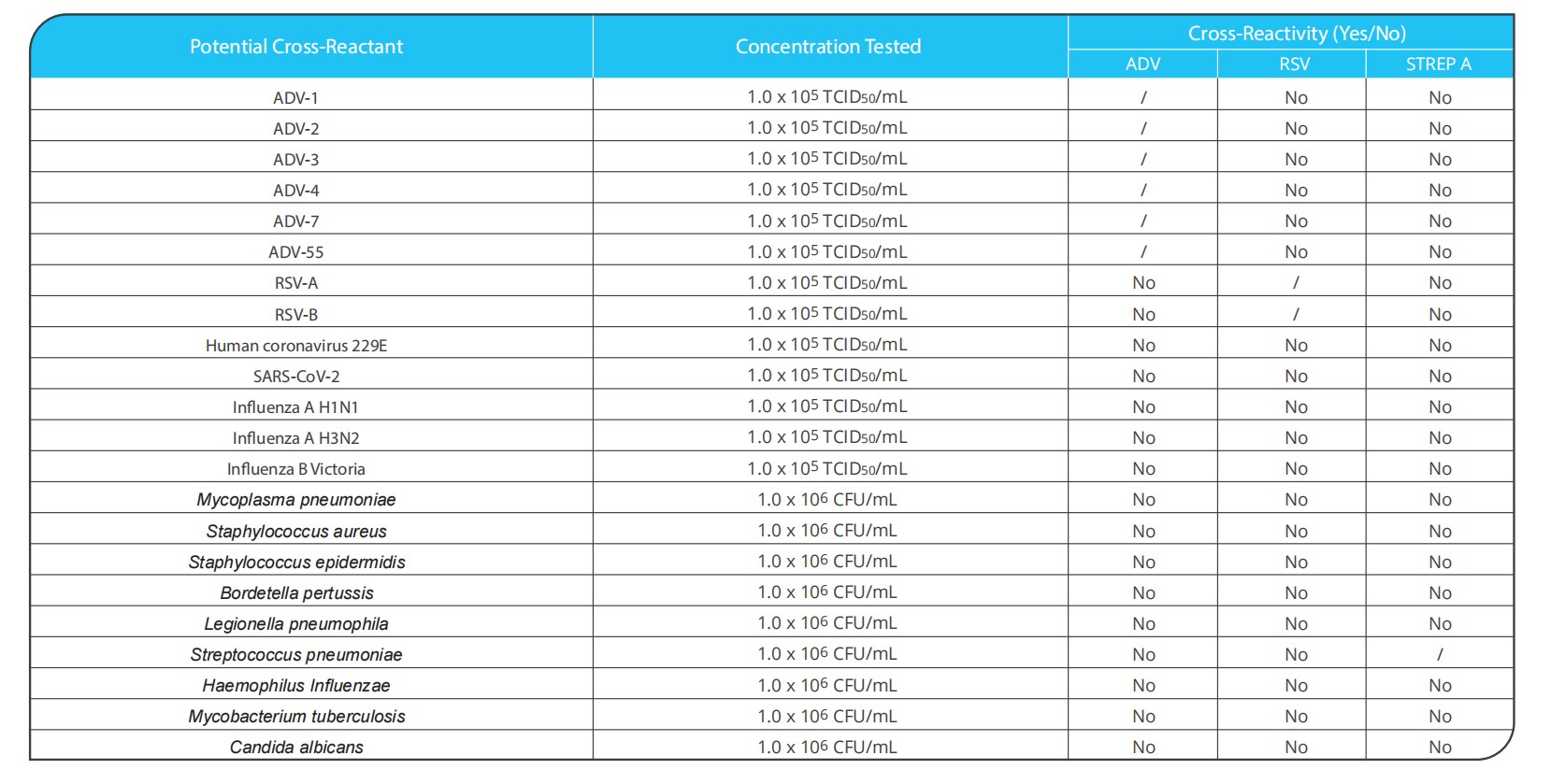
Human adenovirus(ADV) belongs to the adenoviridae family, mammalian adenovirus genus, which is a double-stranded DNA virus without an envelope, mainly infects human respiratory tract, digestive tract and urogenital tract. The main ADV related to respiratory diseases is ADV-B Group (ADV-3, 7, 11, 14, 16, 21, 50, 55), ADV-C Group (ADV-1,2,5,6) and ADV-E group (ADV-4). Acute respiratory adenovirus (ADV) infection which is one of the most common acute respiratory infections in infants and young children. It mainly causes fever, cough, dyspnea and other symptoms.
Respiratory syncytial virus (RSV) belongs to Pneumovirus of Paramyxoviridae with only one serotype, which is a single stranded negative-strand RNA virus with an envelope. RSV infection mainly causes bronchiolitis and pneumonia in infants under 6 months of age and upper respiratory tract infections such as rhinitis and colds in older children and adults.
Group A streptococci, STREP A, is called streptococcus pyogenes, which belongs to β hemolytic streptococcus. Common mode of transmission is spread through the respiratory tract. STREP A can invade any part of the human body, but upper respiratory tract infection is the most common, followed by skin and soft tissue infection. STREP A can cause both suppurative diseases and non- suppurative complications. STREP A is also an indirect cause of the allergic diseases rheumatic fever and acute glomerulonephritis.
Test Principle
Medomics ADV/RSV/STREP A Antigen Combo Rapid Test Kit(LFIA) uses double antibody sandwich method to detect the Adenovirus(ADV), Respiratory syncytial virus (RSV) and Group A streptococci (STREP A) by colloidal gold immunochromatog raphy technology.
When the appropriate amount of test samples treated with lysis buffffer is added to the sample well of the test cassette, the sample will move forward along the test strip by capillary action. If the sample contains ADV, RSV and STREP A antigen, and the concentration is higher than the limit of detection, the antigen will form immune complexes with corresponding antibody labeled with colloidal gold respectively, which are captured by lines A line, R line, and SA line. If test sample contains ADV, RSV and STREP A, forming a red A, R and SA line, indicating a positive result for ADV, RSV and STREP A.
Additionally, the test strip also contains a control line (C line). The C line should be formed to indicate that the sample has been transported properly through the membrane regardless of whether sample contains antigens or not. If the C line does not appear, it indicates that the test result is invalid and the sample need to retest.
Storage Instructions
The test kit should be stored away from direct sunlight at 2℃ to 30℃ with a shelf-life of 24 months. Do not freeze.
Sample Requirements
One test cassette can only be used to test one sample type. Sample types include nasopharyngeal secretion and throat secretion.
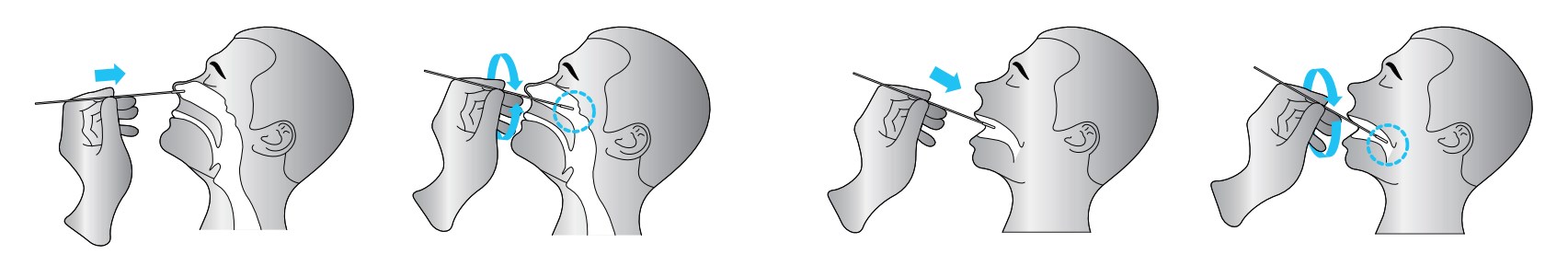
• Nasopharyngeal secretion collection: Take out a swab from the pouch. Insert the swab into one of the patient’s nostrils until it reaches the posterior nasopharynx where is the most secretion, gently rotate and rub the swab over the surface of the posterior nasopharynx for several times before taking it out.
• Throat secretion collection: Insert the whole swab completely into the throat from the mouth, centering on
the throat wall and the reddened area of the palate tonsil, wipe both sides of the pharyngeal tonsil and posterior
pharyngeal wall with moderate force. Try to avoid the tongue before taking it out.
Testing Procedure
Do not open pouch until ready to use. Prep necessary materials: Timer | Tube rack for sampling tubes and specimens | Any necessary personal protective equipment.
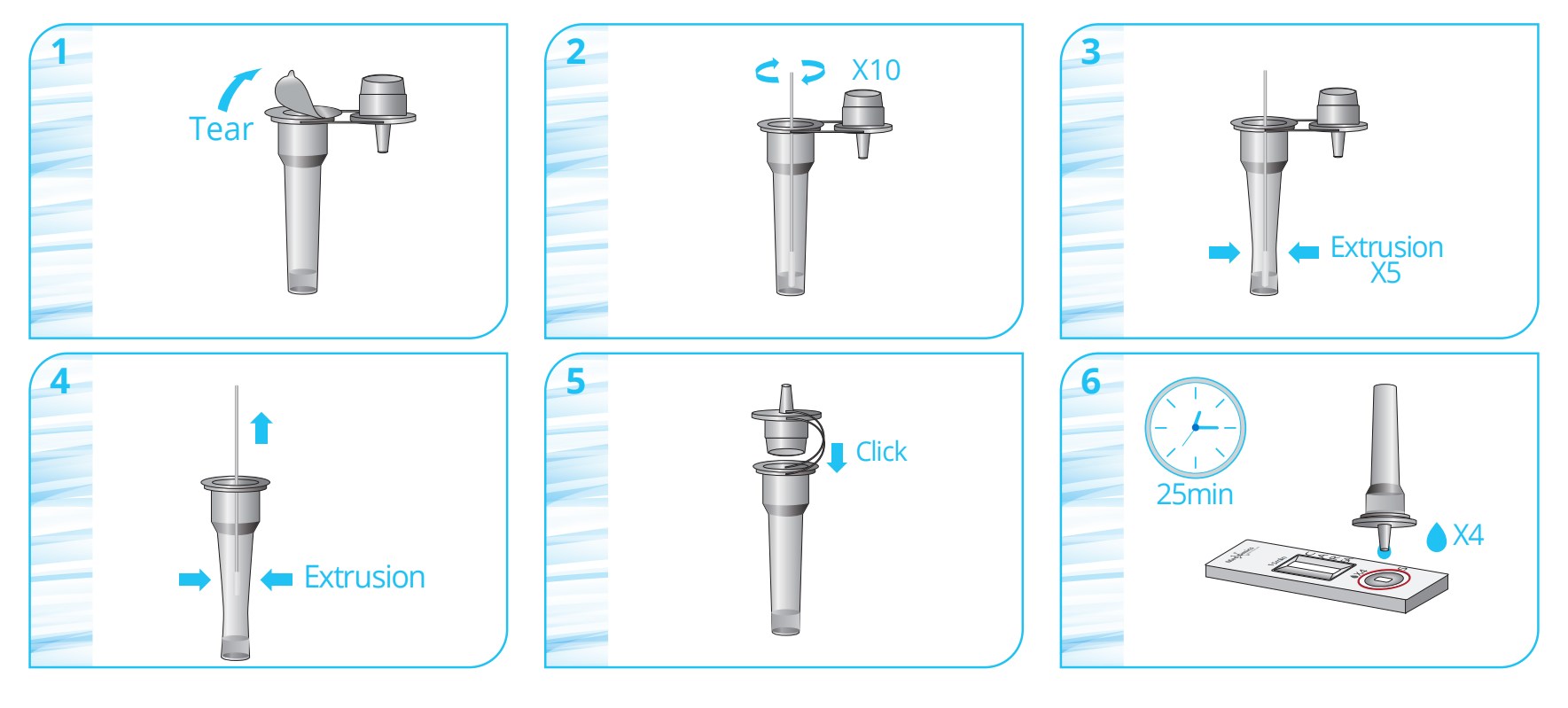
1 | Sampling: Open the seal of the sampling tube containing lysis buffffer. Insert the swab (after collection) into the buffffer. Rotate the swab against the inner tube wall 10 times and squeeze the swab from the outer tube wall 5 times to completely dissolve the sample in the buffffer, then move the swab up until it is resting on the sample solution, squeeze the swab from the outer tube wall in order to leave the sample in the tube as much as possible. Remove and discard the swab, cover the tube with the dropper.
2 | Test procedures: Open the aluminum foil pouch, take out the test cassette and lay it on a clean flflat surface, then mark the cassette with the patient ID or sample number and add 4 drops (approximately 100 μL) processed sample extract into the sample well.
The result should be observed within 20-25 minutes. Results observed after 25 minutes are invalid.
Test Method Limitations
• The accuracy of the test is dependent on the quality of the sample. Improper sampling or storage, using expired samples or repeated frozen-thawed samples can affffect the test result. Test results can also be affffected by temperature and humidity.
• Negative results may be caused by low concentration of ADV, RSV, STREP A antigens in the sample and therefore cannot completely rule out the possibility of infection.
•Some medication (e.g. high concentration of over-the-counter or prescription medication ) in the collected samples may interfere with the test result. Please perform the test again if the result is in doubt.
• This product is only for qualitative testing and the specifific concentration of each indicator must be measured using other quantitative methodologies.
• The results of this test are for clinical reference only and should not be the only basis for diagnosis. Results should be used in combination with clinical observations and other testing methods.
Display of Results/Expected Values
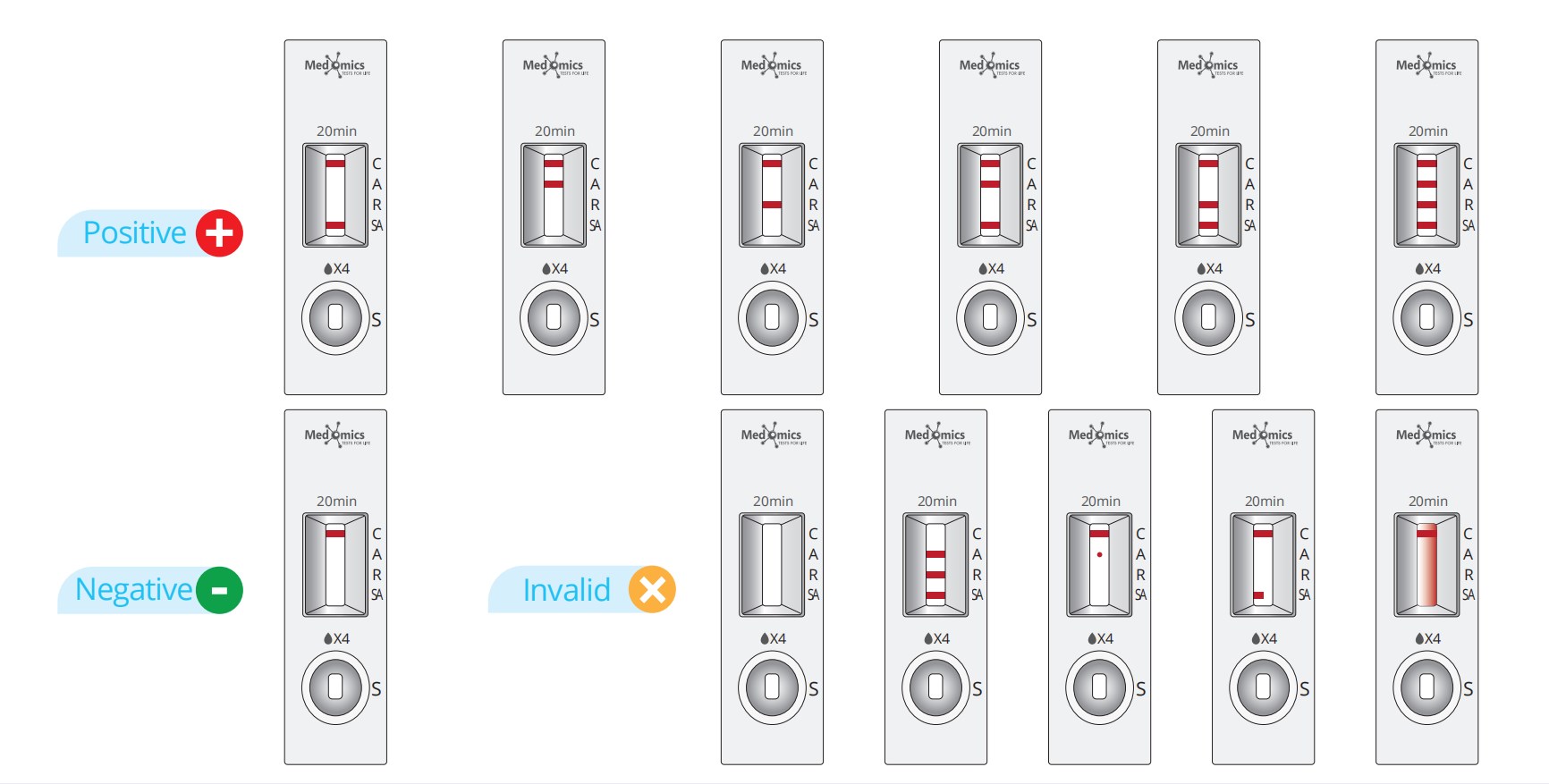
• Negative result: If only the quality control C line appears and the the test line (A line, R line and SA line) is invisible, the sample does not contain ADV, RSV, STREP A antigen or the antigen concentration is lower than the limit of detection, then the result is negative.
• ADV positive result: If the control line (C line) and the test line (A line) appear at the same time, it means that the ADV has been detected and the result is positive.
• RSV positive result: If both the control line (C line) and the test line (R line) appear at the same time, it means that RSV antigen has been detected in the sample and the result of RSV is positive.
• STREP A positive result : If both the control line (C line) and the test line (SA line) appear at the same time, it means that STREP A antigen has been detected in the sample and the result of STREP A is positive.
• ADV/RSV positive result: If there are three lines of control line (C line), ADV test line (A line), RSV test line (R line) shown at the same time, it means that ADV/RSV antigen have been detected in the sample.
• Invalid result: If the C line does not appear, the result is invalid and a new test must be performed again.
Note: The intensity of color that the test line area (A line/R line/SA line) shows will vary according to the concentration of ADV antigen, RSV antigen and STREP A antigen. The result should be determined on whether the test line is formed or not, and is irrelevant to the color intensity. Therefore, any intensity of color in the test area (A line/R line/SA line) should be considered positive.
Product Performance