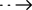Focusing on global epidemic prevention and control, Medomics Antigen Test Products Completed India Registration
- Categories:Newsroom
- Author:
- Origin:
- Time of issue:2021-06-17
- Views:0
(Summary description)
Focusing on global epidemic prevention and control, Medomics Antigen Test Products Completed India Registration
(Summary description)
- Categories:Newsroom
- Author:
- Origin:
- Time of issue:2021-06-17
- Views:0
The Central Drug Standards Administration (CDSCO) of India recently approved the SARS-CoV-2 antigen Test Kit developed by Jiangsu Medomics Medical Technology Co,Ltd. This is the first Chinese company's antigen detection product approved in India.
At the front of epidemic control, the problems of slow and difficult detection are still particularly prominent. It is ,of course ,an ideal state to be able to fully implement nucleic acid detection for all samples, but the requirements for equipment, venues, and professional testing personnel are obstacle to many countries.Moreover, a large number of nucleic acid tests have caused serious medical runs, and it is difficult for medical resources to operate efficiently.
India’s bio-industry is very developed. As of May 25, there are only 9 pure foreign companies (not a Chinese company) among the 42 manufacturers in the whitelist announced on May 25. The rest are local Indian companies or joint ventures, and there are a large number of them. Indian companies under review. The professional version of the antigen and antibody detection kit developed and produced by Medomics has passed the CE certification of the European Union earlier, and has been continuously supplied to many countries and regions around the world, and the antigen detection self-test product has also obtained Germany The Federal Drug and Medical Device Administration approved it as a home self-test product (). As the first Chinese company approved by the Indian Medical Research Council this time, it fully demonstrates that the sensitivity, specificity, stability and other indicators of its products are excellent for the new coronavirus containing Indian variants, and it can be recognized by foreign professional institutions again.

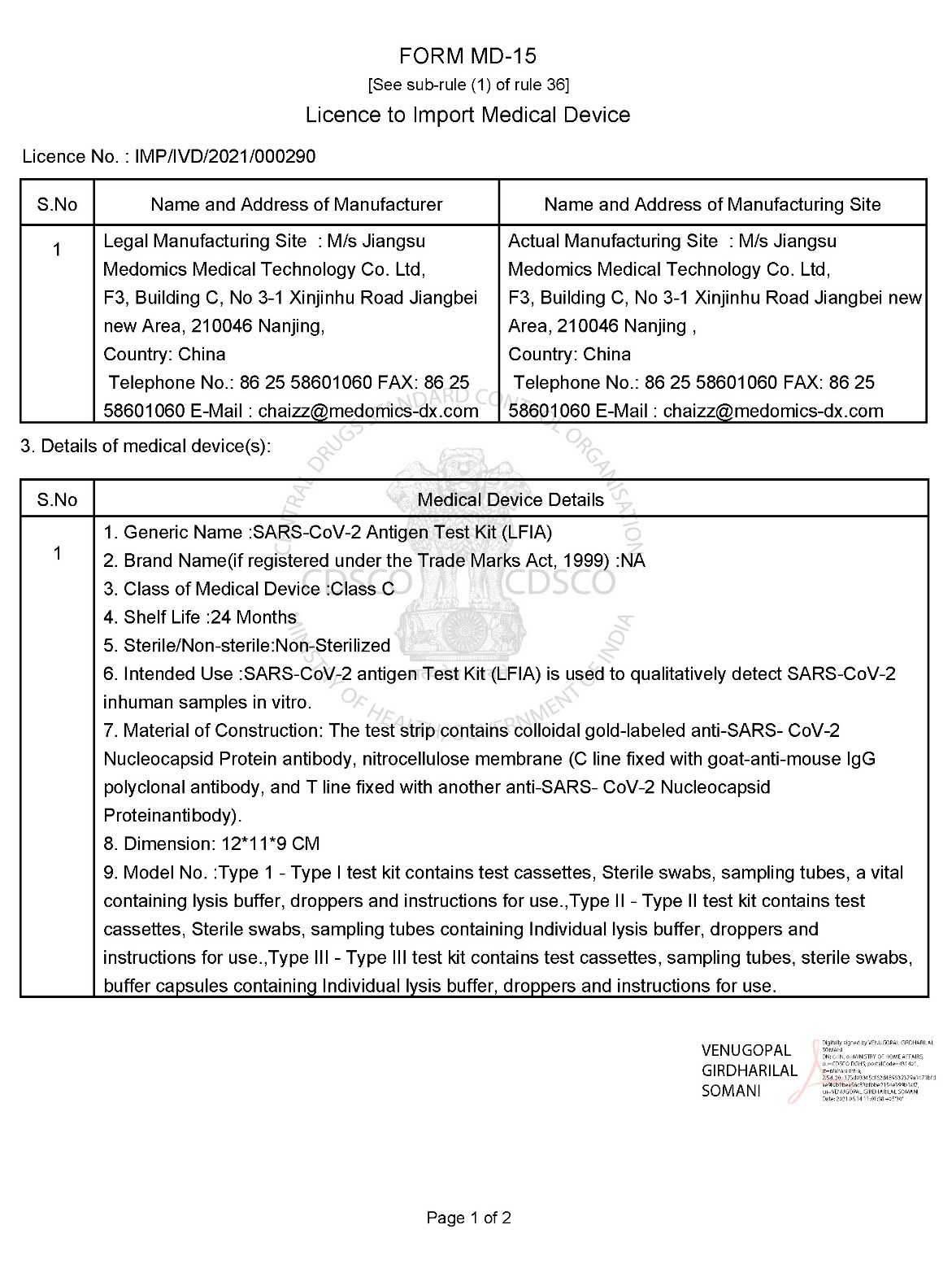
SARS-CoV-2 antigen test kit has the advantages of high accuracy of detection results, simple operation and small size and easy to carry, which is suitable for detecting Coronavirus in various scenes.This has not only greatly saved time and made everyone's life more convenient, but also eased the pressure on hospitals and other testing institutions and contributed to the prevention and control of the epidemic in all countries.
Medomics Medical emerged from the early stage of the COVID-19 outbreak last year with its strong research and development strength and keen market sense, showing the momentum of advancing waves.In the early days of the outbreak, Medomics Medical, as one of the first companies to develop rapid detection reagents, was once listed on Weibo and Baidu's hot search list, and ranked first in the search page of "New Coronet Rapid Detection Reagents" on Google.In cooperation with the State Key Laboratory of Respiratory Diseases, the company has published its first relevant paper on an international journal. In 2020, its products have passed the tests of China Disease Control and Prevention, Harvard Medical School, Columbia Medical School and other institutions.
At present, relying on the strength of excellent R & D center and strict quality management system, the company has passed ISO 13485, Europe CE, Germany BFARM and other authority certification.


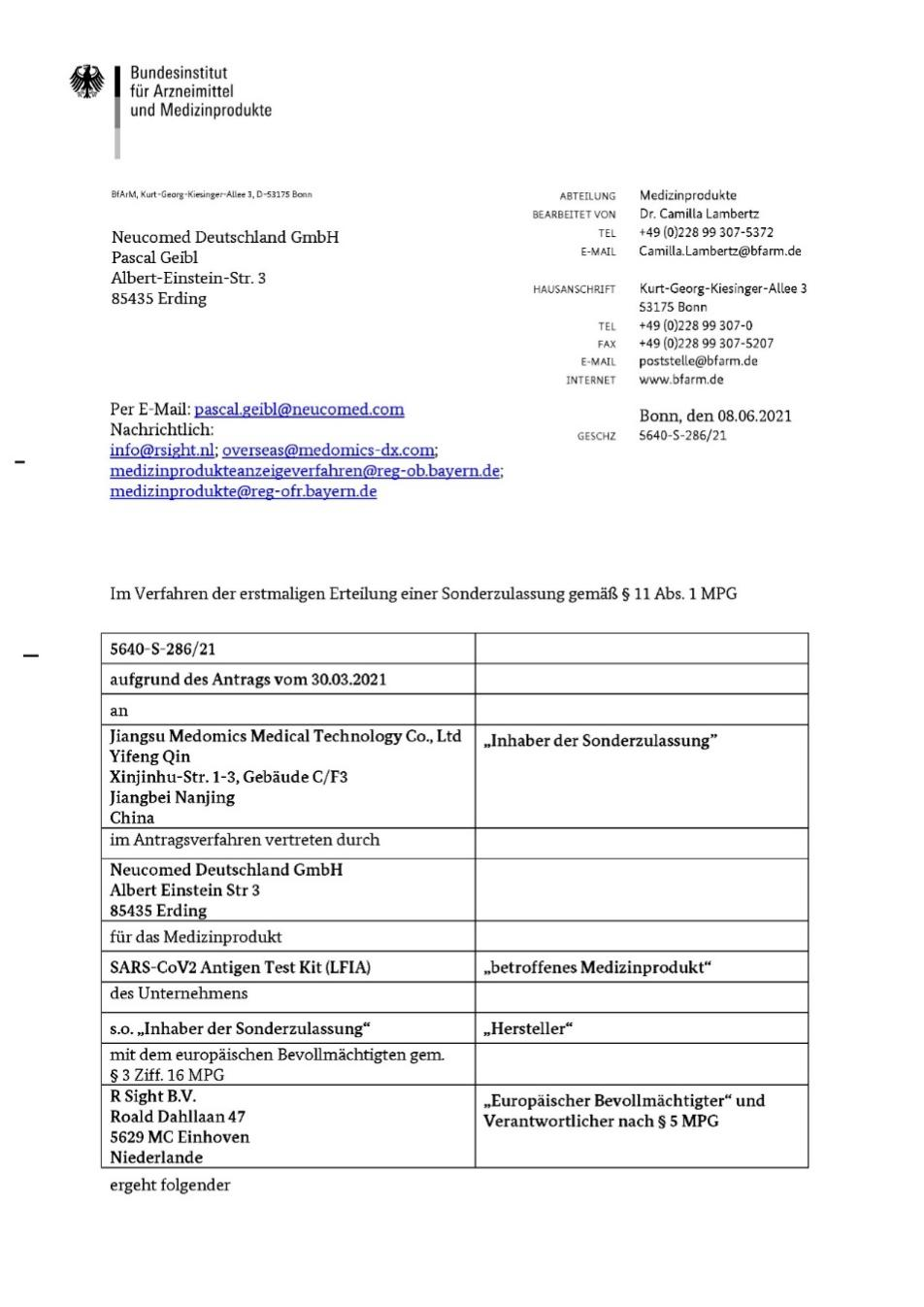
Rapid antigen detection, as a fast and convenient novel coronavirus detection method, can be used as the first line of defense for the detection of early infected persons to assist the prevention and control of the epidemic and control the transmission of the virus.Novel coronavirus will continue to be popular in the long term and the demand for testing will increase dramatically. The approval of Medomics Medical's antigen testing product to enter the Indian market is certainly great news.In the current situation of the epidemic is still very serious, we hope that there will be more products to facilitate the rapid screening and antibody testing of Novel Coronavirus and contribute to the fight against COVID-19.
In the global registration of Medomics excellent consulting companies also played a very important role, such as the Indian registration of Shanghai Maixiang gave a lot of help.
Scan the QR code to read on your phone
- Email Us overseas@medomics-dx.com
- Call Us +86-025-58601060
- COVID-19 Solution
- Top
Contact Us
Phone: (+86) 025 - 58601060
E-mail: overseas@medomics-dx.com
Address:Building 01, Phase 6, No.71, Xinghui Road, Jiangbei New Area, Nanjing
- Email Us
- COVID-19 Solution
- Call Us (+86) 025 - 58601060
- top
Jiangsu Medomics Medical Technology Co,Ltd Powered By www.300.cn